 Aspartame Dangers will shock you!
Aspartame Dangers will shock you!
Do you drink a diet coke or other diet sodas? How many sugar free yoghurts do you eat? Read Aspartame Dangers and be shocked. What about aspartame flavoured cereal, chewing gum, puddings, cakes, juices or one of the other 6,000 products that contain aspartame? Aspartame Dangers will go into how this chemical affects your body. If you have time, take a look at this video: (See Part 2 and Part 3 of this trilogy).
NutraSweet and others
Aspartame is commonly known as NutraSweet, Equal, Spoonful, Equal Measure etc. You will be shocked at how this poison was passed initially by the FDA (Food and Drug Administration). Now it is consumed by millions but will you keep using it after reading  this post?
this post?
But first, I want to relay to you how aspartame was passed as safe by the FDA. This was without proper research, with fraudulent and misleading studies by the then aspartame producing company G D Searle. As well as this, political misdemeanours abounded by Donald Rumsfeld, the CEO of Searle at the time of FDA approval.
How it started
The story starts way back in 1965, when a sweet substance was discovered, quite by accident, by James M. Schlatter, a chemist working for G D Searle & Company. He was working on an anti ulcer drug. After some of the powder was spilt he licked his fingers to pick up a piece of paper and noticed a very strong sweet taste…
A long story
The story of aspartame is a long one, too long to cover in this article so I will try and give you the salient points. This will allow you to make an informed decision as to whether you wish to continue consuming it.
 A good place to start
A good place to start
In the spring of 1971 a neuroscientist Dr John Olney was studying MSG (monosodium glutamate). He informed Searle that his studies had revealed that aspartic acid, one of the ingredients of aspartame, caused holes in the brains of infant mice. (Dr Olney was responsible for having msg removed from baby foods). One of Searle’s researchers, Ann Reynolds, confirms Olney’s findings in a similar study.
Controversial studies
In November 1971, Searle started a 115 week study (No. E-77/78) to test aspartame with 360 weanling albino rats, 190 of each sex. They also did 2 other studies using mice and it was these particular studies that caused much controversy.
FDA approved aspartame
The FDA approved aspartame in July 1974 for limited use as a sugar substitute for sweetening hot beverages, cereals, gum and dry bases. It was not approved for baking goods, cooking or carbonated beverages. FDA scientists found serious flaws in 13 tests related to genetic damage which was submitted by Searle. So why did they give their approval?
A formal objection
in August 1974, a formal objection was filed. Dr John Olney, James Turner and Label Inc. (legal action for buyers’ education and labeling) expressed serious concerns They stated that they believed that aspartame could cause brain damage. Aspartame’s effects on children was of particular concern to them.
Concerns over other drug studies
 We’re only talking about aspartame here. But in July 1975 the FDA commissioner Dr Alexander Schmidt appointed a Task Force to look into 25 studies for the drugs Flagyl, Aldactone, Norpace and Aspartame. This was because of
We’re only talking about aspartame here. But in July 1975 the FDA commissioner Dr Alexander Schmidt appointed a Task Force to look into 25 studies for the drugs Flagyl, Aldactone, Norpace and Aspartame. This was because of  concerns of Searle’s responses to queries about the testing of their drug Flagyl. Also there were serious and unexpected side effects from other drugs Searle developed. The Olney studies and information, started controversy within the FDA as to the
concerns of Searle’s responses to queries about the testing of their drug Flagyl. Also there were serious and unexpected side effects from other drugs Searle developed. The Olney studies and information, started controversy within the FDA as to the  validity of Searle’s research of aspartame and other drugs. All of the studies were either by Searle or done for Searle, ie. Hazleton Laboratories. Eleven of the studies involved aspartame.
validity of Searle’s research of aspartame and other drugs. All of the studies were either by Searle or done for Searle, ie. Hazleton Laboratories. Eleven of the studies involved aspartame.
Approval put on hold
In December 1975 there was a hold of approval by the FDA for aspartame, due to findings of the Task Force. The evidence of the aspartame pivotal studies were protected under FDA seal on December 3rd 1975.
Searle building an aspartame facility
Meanwhile, Searle was building a production facility for aspartame and had so far spent 19.7 million dollars. On 8th December 1975, stockholders filed a class action lawsuit against Searle. They accused them of concealing information regarding the quality of research at Searle. This being in violation of the Securities and Exchange Act.
 500 page report completed.
500 page report completed.
In March 1976 a 500 page report by the FDA Task Force was completed. Here are some of the conclusions from this report. Searle’s research practices have been taken from Mark D. Gold’s FDA Dockets Submittal which has all relevant references therein.
“We have noted that Searle has not submitted all the facts of experiments to the FDA, retaining unto itself the unpermitted option of filtering, interpreting and not submitting information which we would consider material to the safety evalution of the product….Finally we have found instance of irrelevant or unproductive animal research where experiments have been poorly conceived, carelessly executed, or inaccurately analyzed or reported.”
and
“Some of our findings suggest an attitude of disregard for FDA’s mission of protection of the public health by selectively reporting the results of studies in a manner which allay the concerns of questions of an FDA reviewer.”
 Worrying research practices
Worrying research practices
“Excising masses (tumors) from live animals, in some cases without histologic examination of the masses, in others without reporting them to the FDA.” When Searle’s representatives were questioned they stated “these masses were in the head and neck areas and prevented the animals from feeding.”
“Failure to report to the FDA all internal tumors present in the experimental rats, e.g., polyps in the uterus, ovary neoplasms as well as other lesions.”
“Instead of performing autopsies on rhesus monkeys that suffered seizures after being fed aspartame, the company had financed a new monkey seizure study with a different methology that showed no problems.”
Animals which had died were sometimes recorded as being alive and vica versa. “These include approximately 20 instances of animals reported as dead and then reported as having vital signs normal again at subsequent observation periods.”
“Never seen anything as bad as Searle’s studies!”

Phillip Brodsky described the 1975 FDA Task Force members as some of the most experienced drug investigators. Brodsky was the lead investigator. He went on to state that he had never seen anything as bad as Searle’s studies.
The FDA Commissioner at the time, Alexander Schmidt stated “[Searle’s studies were] incredibly sloppy science. What we discovered was reprehensible.”
Senator Kennedy says “profoundly disturbing.”
Senator Edward Kennedy at the April 1976 hearings, before the Senate subcommittee on Labor and Public Welfare stated: “The extensive nature of the almost unbelievable range of abuses discovered by the FDA on several major Searle products is profoundly disturbing.”
 An independent study
An independent study
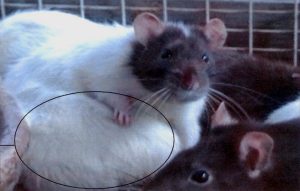
To finish Part One of this post, take a look at this research. This was done by an independent researcher who just wanted to know the truth. Victoria Inness-Brown started the study because of concerns for her family who drank many diet sodas.
Victoria came across ‘The Bressler Report’ which we will discuss in Part 2. Hence, she decided to do her own experiment so she could see for herself if aspartame did actually produce tumours. She used rats for the study, 108 of them that she raised herself. Victoria had a control group as well as an experimental group.
NutraSweet used in study
 She could not obtain the pure form of aspartame because she wasn’t a food manufacturer consquently she used NutraSweet which she put into the rats’ water. The rats were given the acceptable daily limit or acceptable daily intake (ADI). This is set by the FDA which is 50mg per kilogram. The research took 2.5 years, ie. 30 months to complete. Also, she had no help with finance but stated that it wasn’t too expensive to do the study, just time consuming. She did all the care for the animals and was meticulous with recording her research.
She could not obtain the pure form of aspartame because she wasn’t a food manufacturer consquently she used NutraSweet which she put into the rats’ water. The rats were given the acceptable daily limit or acceptable daily intake (ADI). This is set by the FDA which is 50mg per kilogram. The research took 2.5 years, ie. 30 months to complete. Also, she had no help with finance but stated that it wasn’t too expensive to do the study, just time consuming. She did all the care for the animals and was meticulous with recording her research.
As you can see by the images here, the results of the study are quite horrendous. If you wish to look at more information regarding this study you can go to her page at www.AspartameExperiment.com. or check out the narrative of her special interview with Victoria Inness-Brown and Dr Mercola.
 In Part 2 of this article, the Bressler Report is instigated by the FDA and Donald Rumsfeld joins G D Searle. President Ronald Reagan comes to power in 1981. Approval of aspartame comes soon after.
In Part 2 of this article, the Bressler Report is instigated by the FDA and Donald Rumsfeld joins G D Searle. President Ronald Reagan comes to power in 1981. Approval of aspartame comes soon after.
Spread the word!

I would go through periods, especially when work traveling, drinking diet coke from convenience stores. When I went through these periods, I would experience muscle spasms in my forehead, above my right eye, chest arms and legs. When I stopped drinking diet soda, the spasms would stop.
In 2000, in my 30s, I was going through a period of drinking diet soda and eating less. On a Saturday afternoon, while stepping from my car into the cold weather, I experienced chest pain and eventually diagnosed a Torsades des Pointes cardiac arrhythmia but the cause was never determined, though possibly a coronary spasm. I am convinced that aspartame was a contributor. I was also hypokalemic and hypomagnesic.
The combination of caffeine, aspartame, and the dehydration that resulted in low magnesium nearly killed me.
Hi Jeff,
Thank you so much for writing about your experiences over the years. You certainly have gone through the mill! It is so important to impart this information. Others in a similar position to you can learn much from your comments and it could even save lives and drastically improve health. To learn exactly why you are ill is very liberating, especially when you know you can change your life by taking charge of your own health. Good luck to you and I’m sure your health, now it is in your hands will improve.
At breakfast this morning, my husband (a “foodie”) was closely reading the Log Cabin Syrup Sugar Free label and said this has Aspertame in it!! Knowing nothing about it, I immediately googled it. When I read “Aspertame dangers — this will shock you” under magnesiumandhealth.com I was horrified when I read it and MORE IMPORTANTLY that it is STILL being found in food products in 2022!!! Ironically, the label says “CAUTION: DO NOT MICROWAVE THIS BOTTLE” when the product inside can do your body so much damage!
Thanks for your comment Kathryn. I’m so pleased you have taken the article on board. It is indeed a horrid product and should be taken off the market. But you know what happens, follow the money! At least you know to steer well clear of this artificial sweetener. Good health to you and your family!
wow, Thanks for this amazing write up, it is quite revealing, no wonder you won’t find any of this in any publication despite the damage such products are causing. I guess there is a lot of politics and conspiracy behind the scenes. This article is a serious wake up call for me, never again will will I consume any of those aspartame laden sweets and food/snacks; a word is enough for the wise. But the serious question on my mind now is that if the FDA knows about these deadly products containing aspartame being sold all around us why then are they still being sold .
Hi there Zuchii and thanks for your input. I’m pleased you’re more aware now that you may be consuming a product that could do harm to you. In fact, there are many so called foods out there that we should steer clear of. The trick is knowing which they are. With aspartame, it should be displayed on the package, so it can easily be seen.
The FDA are in the pockets of the pharmaceuticals and chemical industries and some members of the FDA, can be persuaded to do their bidding. They will often take research from pharmaceuticals at face value without checking for fraud and often turning a blind eye. You have to be in charge of your own health nowadays and do your own research to have a chance of staying healthy.
This is very disturbing. It makes you think how much money does the food industry make to get all these poison passes and used in our food. I always stay clear from all this sweetener like aspartame and Sucralose. First of all they have the after taste that I hate and second of all who knows what will actually happen to my body, since each individual reacts to each chemical differently. It could be fatal to me who knows, NO THANK YOU.
When I buy food, I always be mindful of the additives. If the additives is natural and something that I can pronounce, I am most likely will buy it. I rarely buy packaged food, but since I am not perfect, I do buy them sometimes but it is very minimal. Also, if the ingredients has more than 5, I stay clear from that as well. What I actually do now is I bake my own bakery and make my own chocolate, since it has less chemicals. If I crave soda, I will mix club soda or seltzer with my own fresh squeeze juice as well. It takes time to get use to the taste but after a while you will enjoy it.
Let’s us all be aware of what we eat, and stay healthy together. Great article.
Hi Nuttanee and thanks for your comments. Yes, it is disturbing and the deeper you go into the modus operandi of the food companies, the pharmaceuticals and the chemicals companies, the worse it gets. You have to be aware that it is only you that will after your health. Most medics have to do as they’re told by the pharmaceuticals. If they don’t tow the line then they will be vilified or even have their licence pulled.
Keeping clear of processed foods as well as additives is a good move. Checking the ingredients is not always easy as those classed as GRAS (generally regarded as safe) can be left off the labels. As you’ve probably guessed, they are not always safe, it is a loophole.
We must be diligent and do our own research. There is nobody out there looking out for you, even most doctors are under the thumb of the pharmaceuticals. As you say, question everything and be aware at all times.
Wow, it’s crazy how dangerous modern day foods can be. This is going to stop me from drinking any soda for a while. I’m just wondering though, I may have missed it in the article, is Aspartame still being used in diet sodas? It seems to be that your report is rather based in the late 1900s.
Well done Kevin, keep away from aspartame. You can check if it’s in your food or drink as it has to be displayed on the label, for the moment anyway. They are trying to get the labelling off of milk though, so look out for that one! Aspartame has been around for a long time but it is not generally recognised as being the danger that it is. Clever marketing and advertising I suppose?
The information regarding aspartame being found in foods is alarming. I heard about sweeteners in sodas not good for you as well as msg being found in packaged seasonings but I had no idea that those were found in cakes too. Wow! I just think that people should do research before they decide to buy them. Thank you so much for sharing!
Hi Carmeta and thanks for your input. It is unfortunate that in this day and age, money is king and all else seems to go out the window, including ethics. The government and their agents, pharma, food industry and chemical industry are in it just for profits. You and your health is unimportant to them. So, that means you have to take the reins and look after yourself and your family. Looks like you know that already!
What an incredibly informative article. I have heard or dangers or just that it might not be that great for your body but I had no idea the depth of harm that this chemical can actually do. Very sad to see outside confirmation with these poor animals. This makes me wonder what all I eat/drink that actually has aspartame in it, looks like i will be cutting out some products from my diet.
I’m pleased you found the post informative Alisha. Now you can act on it and protect yourself and your family from this poison. If you do need to use a sweetener, Stevia seems to OK with no adverse affects reported so far. Aspartame was approved fraudulently which proves if they had to go to that length to get it passed, they knew it was toxic. Just shows, these large consortiums have one aim only, to make money, even if it’s at the expense of your health.
The information regarding aspartame being found in diet sodas, cakes and other foods is alarming. I heard about sweeteners in sodas not being good for you and as well as msg being found in packaged seasonings but I had no idea that it is also found in cakes. Wow! I just think that people need to do research on these things before they decide to buy them. Thank you so much for sharing!
Fortunately, I stopped using Aspartame a long time ago but this information is till shocking considering how many years I used it unsuspectingly. not to mention many family members and friends that continue to use it right now.
As a diabetic, I can’t do regular sugar. I use Stevia and hear good things about Monk fruit extract, but have not tried it.
Thanks for your comment Chazz. Aspartame is often taken by diabetics and understandably so. They think they’re doing the right thing and that’s not their fault. It’s misinformation or shall I say hidden information by the big pharmaceuticals. They don’t want you to know what aspartame actually does to you, it would impact on their business! I think we forget that big pharma are companies out to make profits, that’s what companies try to do. The fact that what they do impacts on our health, is something they don’t seem too concerned about. I’m so pleased you found out about this chemical concoction a long time ago. Good health to you and yours, Ches
Thank you so much for this informative article. I have been using Splenda in my iced tea, thinking that this sweetener was superior. Now that I am aware, I can try to use Stevia instead, although the taste is a little different. It gets tricky because we don’t eat a lot of sugar in my home because my husband is a diabetic. Good information! Take Care, Heather
Hi Heather, yes I think stevia is the best alternative so far. Try and keep off the aspartame, it certainly won’t help your husband’s diabetes. Apart from the dangers shown by many studies and the problems caused by each of aspartame’s components, using aspartame as a dietary aid is a farce. This neurotoxin actually encourages weight gain. The two amino acids phenylalanine and aspartic acid, stimulate insulin production. This in turn causes spikes in insulin which takes all the glucose from the blood, stores it as fat and leaves the body ravenous. Thus this encourages consumption of more food and the process is repeated; a vicious circle.
I know it’s difficult but please try and adapt, I’m sure you and your husband will feel the benefits. Part 2 of this trilogy comes out tomorrow and part 3 on the 17th April which goes into what happens to the body in more detail. Good health to you and your family. Ches
Hi Chessie.
I sometimes do drink diet sodas; but I’m going to stop. It is not worth the health risk. I have heard so much about aspartame being bad for you. And now your post confirms that it is a health risk. I think you are doing a great service by advising people of the risks they take when they consume this product.
What do you know about Splenda? Is it safe? How about Stevia? Or do I just have to stick to plain sugar to be safe?
Jeannie
Hi Jeannie and thanks for the comment. Splenda is a chlorinated artificial sweetener in line with aspartame and saccharin, and with detrimental health effects to match. Stevia seems to be OK and I would use it if I had to. I don’t use sugar at all but always look for sugar in anything I buy instead of any other sweetener. I’m really pleased you’re kicking aspartame to the kerb. It is rather addictive and many people have trouble giving it up, I’m sure you will not be one of those. Good health to you and I’m sure it will improve now you’re not taking aspartame. Ches
I remember many years ago a lady approached me to fill in her three question survey that was about ‘alternative sugar for kids’. Her survey suggested that aspartame sweeteners were better and good for children.
Question one, she asked me which of these three products would you give your child. lol there was no option to tick ‘none’. However, I said I would not give my children any of those. Question two was the same and my answer was the same. Question three, I remember it being about sodas, but my answer was the same again. She said, your not helpful are you, I need to find someone who’s happy to feed their kid’s aspartame.
Hi Sharon and thanks for your input. The problem is there is so much hype by those who are gaining revenue because of aspartame and they tell all kinds of stories about aspartame being the most researched food item. They tell untruths, and the public is so trusting, they believe everything they say. The only way you can be in charge of your own health is to research what worries you, yourself. You can then become as expert at nutrition, vitamins and minerals as most doctors and probably will be able to teach your doctor a thing or two. The clue is to look for research, studies and information given out by those that do not benefit financially by it, those without an axe to grind. This is much safer than taking notice of big pharma, big agra, governmental bodies and the such like. Good health to you and yours. Ches